 If you follow Rett research closely (or maybe even loosely), this will be a drug name you’re familiar with. My Google analytics tells me it’s one of the most popular searches currently bringing people to this blog so I know you’re out there and looking for information about this potentially groundbreaking new medication and the implications it may have for Rett syndrome.
If you follow Rett research closely (or maybe even loosely), this will be a drug name you’re familiar with. My Google analytics tells me it’s one of the most popular searches currently bringing people to this blog so I know you’re out there and looking for information about this potentially groundbreaking new medication and the implications it may have for Rett syndrome.
NNZ-2566 is a medication being developed by the small pharma company Neuren. Neuren is a company in New Zealand which has offices in Australia and the United States. They have special interests in areas of neuroscience where there are gaps in development of treatments. Neuren currently has a growing portfolio on traumatic brain injury, neurodegenerative disorders and neurodevelopment disorders.
There are very few human clinical trials actually going on for Rett syndrome and NNZ-2566 is one of them. The trials are happening in America as we speak.
[What]
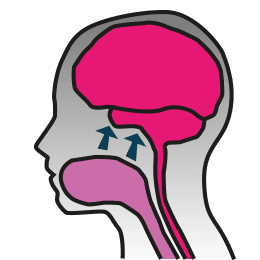
So the first question I aim to answer for you is “what is it?” We have a protein prevalent in our bodies called IGF-1. Its function is to help cells grow. It is an important component in the actions of the growth hormone in all of our bodies. In the brain, it helps cells to divide and make synaptic connections from neuron to neuron. However, people with Rett syndrome have issues with their IGF-1 because MeCP2 affects its expression. Now, where NNZ-2566 comes into this mix is this: by looking at the IGF-1 protein and which bits of the whole package could be beneficial in treating Rett, scientists have found that the first 3 amino acids of IGF-1 are what’s needed.
IGF-1 does not readily pass from our circulation into the brain. However, a small piece of IGF-1, called a tripeptide, is able to pass from the blood and into the brain. NNZ-2566 is a synthetic version of the tripeptide that has been modified so that it can be absorbed orally, passing from the digestive tract into the circulation and then into the brain. Evidence from the laboratory suggests that the tripeptide is the part of IGF-1 that seems to be effective in treating Rett syndrome. A trial of NNZ-2566 is underway in adolescents and adults with Rett Syndrome at Baylor College of Medicine and the University of Alabama. A trial of IGF-1 in young girls is underway at Boston Children’s Hospital.
[How]
NNZ-2566 is delivered as an oral, strawberry flavored liquid whereas IGF-1 has to be injected.
[Contraindications]
Just like any drug, IGF-1 isn’t suitable for everyone. When growth hormones are given after puberty, they can cause continued bone growth so there’s a window of time when this could be administered as a treatment. However, NNZ-2566, which is a peptide and not the whole protein, does not appear to have those same issues. The clinical trial with NNZ-2566 is designed to show that it is suitable for teenagers and adults with Rett. This means NNZ-2566 could have the potential to be suitable for anyone with Rett at any period of their life, something that no other suggested treatment we’ve ever seen can claim.
[The Benefits]
NNZ-2566 is an easy, strawberry flavoured liquid that crosses the brain/blood barrier.
You may remember a while back, I posted about gene therapy and the post said that, while treatment with vectors is an interesting idea, there are great challenges. That these methods were like using a shotgun when what we need is a sniper rifle.
Well, IGF-1 is like a sniper rifle. Through the current trials, scientists are hoping to deliver more growth enhancers into the central nervous system which gets right to the central problem immediately, bypassing the need to deliver a new gene with a vector and hope it is expressed to correct the defective MeCP2.
But of course, I’ve already mentioned that IGF-1 needs to be injected while NNZ-2566 is an oral medication to hopefully deliver the exact same results and be potentially suitable for any age.
[The Challenges]
Like every current suggested method for the treatment of Rett, there are a few challenges to face. The fundamental challenge with NNZ-2566 is that it is new drug, and new drugs have to go through a lot of rigorous testing before they can be used on all age groups. Since NNZ-2566 was developed by Neuren for traumatic brain injury, it has had a sizable amount of safety testing already completed and has been shown to be generally safe and well tolerated by adult human subjects. However, this fact does not remove the need for safety evaluation in Rett patients.
[Current Studies]
NNZ-2566 is currently being studied in human clinical trials for both Rett syndrome and traumatic brain injury. Both conditions have something in common in that there are weak synapses in the brain. A study in Fragile X syndrome is scheduled to begin shortly.
Phase 1 (the safety tests) have been completed. We know that it’s a well-tolerated drug.
Phase 2A began in May 2013 and hopes to end in September this year. These are currently being undertaken at Baylor College of Medicine with Drs. Dan Glaze and Jeff Neul and at the University of Alabama – Birmingham with Dr. Alan Percy. This trial consists of 60 participants between the ages of 16-45.
The Phase 2 study examines the safety profile, looking for unexpected adverse changes, deterioration or anything that wouldn’t be expected in the natural progression of Rett syndrome. This is where the ongoing Natural History Study adds value. Unless we study Rett in the long run and learn what happens and the effects we can expect, it’s difficult to measure the effectiveness of drugs we want to trial for Rett. The data extracted during the Natural History Study gives us baselines of what to expect so that during these types of trials, we can know what would normally have been expected and gives scientists a greater base of knowledge for use when monitoring such trials.
This phase is also measuring efficacy (effectiveness).
Based on the tests with mouse models, we’re hoping to see improvements in breathing and cardiac function, and decreases in seizures. The patients are also being observed for changes in many other behaviors.
[The Next Step]
The next step for NNZ-2566 will be proof of concept studies in the pediatric population. Remember, the youngest person in the current study is 16 years old because the FDA guidelines are such that a drug usually cannot be tested on juveniles until the trial has first been successful with adults (when adult patients are available).
[Funding]
This is where partnerships are incredibly important. IRSF started the Baylor study with a $600,000 ANGEL grant to get it going, but this doesn’t even come close to covering the cost. Partnership with Neuren is extremely important to bring more resources to the table for these trials.
[Considering a Trial?]
It’s very important to fully understand the implications of putting your child into a clinical trial. Please view this document from IRSF on safety in Rett Syndrome trials.
This post was made possible by contributions from:
- Steve Kaminsky, Ph.D. – Chief Science Officer, IRSF
- Janice Ascano, Ph.D. – Manager of Grants and Research, IRSF
- Nancy Jones, Ph. D. – Senior Director, Clinical Development and Medical Affairs at Neuren Pharmaceuticals





